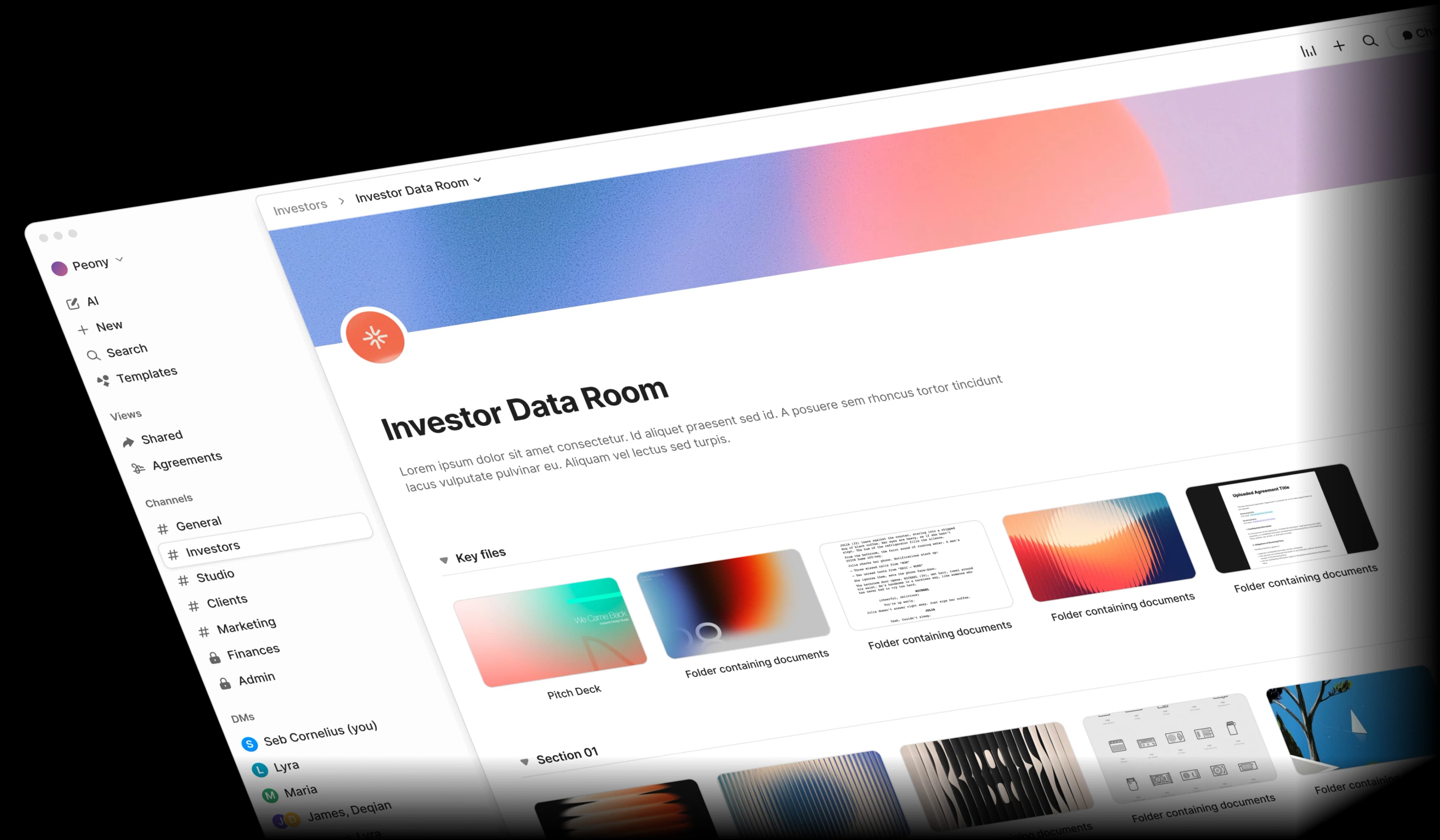
Every site, every protocol, one secure hub.
The data room purpose-built for CROs, trial sponsors, and research institutions. Distribute study protocols across sites, manage IRB submissions, and maintain 21 CFR Part 11-ready audit trails.Set up in under 5 minutes.
No credit card required
Clinical trial document management is broken
Between multi-site coordination, regulatory submissions, and monitor reviews, research teams share sensitive trial documents across dozens of insecure channels. One protocol leak can compromise an entire study.
Multi-site chaos
Distributing protocol amendments, Informed Consent Forms, and Case Report Forms across 20+ research sites via email leads to version confusion and compliance gaps.
Patient data exposure
Aggregate safety data, SAE reports, and DSMB documents contain sensitive patient information. Generic file-sharing tools lack the access controls research demands.
Cross-jurisdiction filings
IRB and ethics committee submissions vary by country. Managing different document packages for FDA, EMA, and local boards across scattered tools is error-prone.
Protocol version control
Study protocols go through dozens of amendments. Without proper version tracking and distribution logs, sites may operate on outdated procedures.
Built for how clinical research teams actually work
Peony gives you controlled document distribution, 21 CFR Part 11-ready audit trails, and AI-powered research intelligence — all at a fraction of legacy VDR pricing.
Personalized links, watermarked protocols, and NDA-gated access
Distribute Study Protocols, Investigator Brochures, and Informed Consent Forms to every research site with unique personalized links. Watermarks trace any leaks back to the source. External monitors and CROs sign NDAs before accessing trial documents.
Personalized links per site
Send each research site a unique link to protocols and CRFs. Track which sites have accessed the latest amendments and which are still reviewing outdated versions.
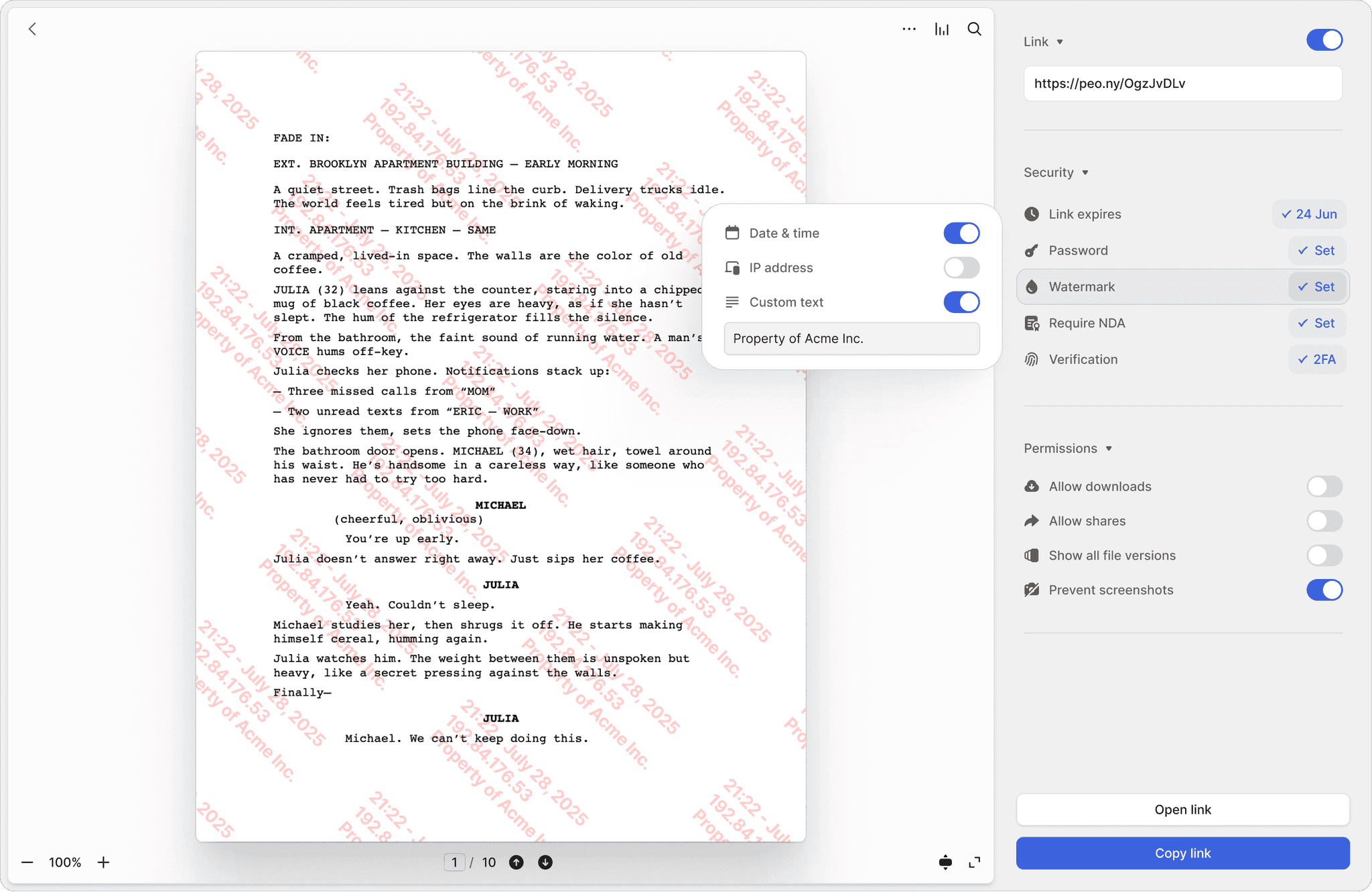
Watermarked trial documents
Every page of your Study Protocol and Investigator Brochure is stamped with the viewer's identity. If a confidential protocol leaks, trace it instantly.
NDA-gated monitor access
Require external CROs, clinical monitors, and site auditors to sign an NDA before viewing any trial data. Fully integrated e-signatures included.
21 CFR Part 11-ready audit trails for every document
Track every view, download, and signature with timestamped logs. Two-factor authentication protects access to patient data summaries and DSMB reports. Page-by-page analytics reveal exactly which protocol sections reviewers focused on.
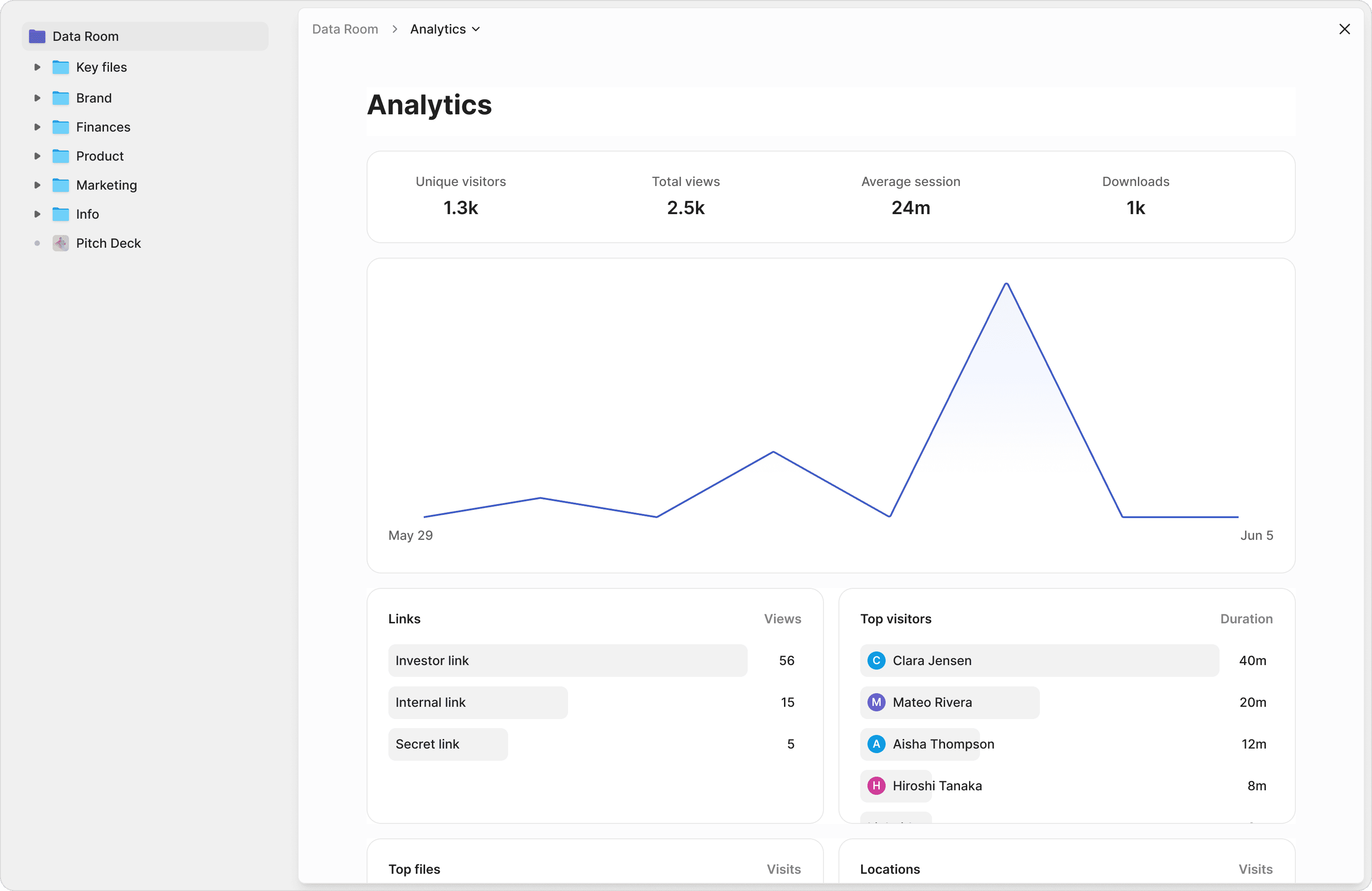
Page-by-page protocol tracking
See which sections of your Study Protocol or SAE reports IRB reviewers and monitors spent the most time on. Identify potential concerns before they become queries.
2FA for sensitive data access
Require two-factor authentication before anyone can access patient data summaries, DSMB reports, or unblinded safety data. Exportable logs for regulatory inspections.
Granular access by role
Give principal investigators full protocol access, monitors read-only CRF access, and ethics committees access to specific submission folders — all from one data room.
Search across protocols, brochures, and regulatory submissions
Stop digging through hundreds of trial documents. Ask natural language questions across your Investigator Brochures, Study Protocols, and regulatory submissions to find specific endpoints, inclusion criteria, or safety data in seconds.
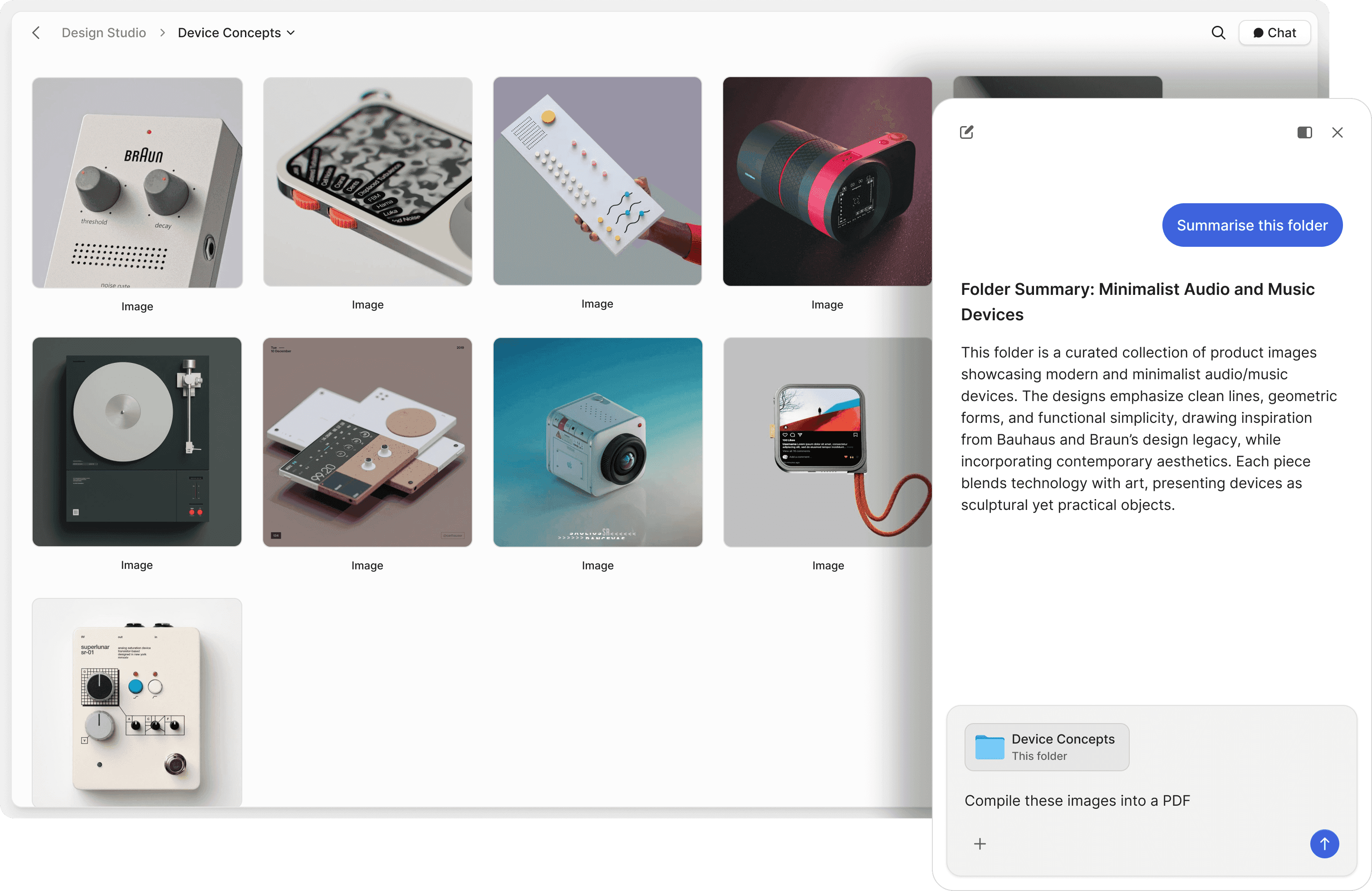
Chat with trial documents
Ask questions across Study Protocols, Investigator Brochures, and DSMB reports. AI retrieves answers with exact source references from your trial master file.
Find endpoints and safety data
Search your data room in plain English. Find specific primary endpoints, inclusion/exclusion criteria, or adverse event data without remembering file names or section numbers.
Monitor Q&A on autopilot
Let AI answer clinical monitor questions about protocol procedures, eligibility criteria, and study timelines 24/7. Track what they ask to anticipate site queries.
"Peony has been great for sharing documents with investors, employees, and customers. It's easy to use, good value, and new features are constantly being added. Definitely recommend!"

Ed Harris
Founder & CEO, Ligo Bio (YC S24)
Panos Moutafis
Founder & CEO, Zenus
"Every now and then, you come across a product that has a massive positive impact on your business. Peony is one such product."
Frequently asked questions
What is a clinical research data room and how do CROs use it for trial document management?
A clinical research data room is a secure virtual environment where CROs distribute protocol amendments, manage monitor access to case report forms, and maintain the trial master file across multiple research sites. Peony provides a purpose-built clinical research data room with personalized links per site, screenshot protection, dynamic watermarks, and 21 CFR Part 11-ready audit trails — so a CRO can onboard a new multi-site trial without waiting on IT, all deployable in under 5 minutes.
How do CROs securely share trial documents across multiple research sites?
CROs need to distribute protocol amendments, case report forms, and safety data across dozens of sites without version confusion. Peony solves this with personalized links per research site, so you can track exactly which sites have accessed the latest amendments. Unlike legacy VDRs like Ideals or Firmex that take weeks to configure, Peony deploys in under 5 minutes with granular per-site permissions.
Does Peony support 21 CFR Part 11 compliance for clinical trials?
Peony provides 21 CFR Part 11-ready audit trails that log every view, download, and signature with timestamps and user identity, backed by AES-256 encryption at rest and TLS 1.3 in transit on SOC 2-ready infrastructure. Peony is GDPR, CCPA, and HIPAA compliant. Two-factor authentication protects access to sensitive patient data summaries and DSMB reports. These exportable compliance logs make Peony suitable for FDA-regulated clinical trial environments where legacy VDRs like Firmex charge significantly more for similar capabilities.
How does Peony protect patient data and sensitive safety reports?
Peony protects patient data through multiple layers: screenshot blocking prevents unauthorized captures of aggregate safety data, dynamic watermarks trace any document leak back to the specific viewer, and NDA gating requires external monitors and CROs to sign agreements before accessing any trial materials. Peony's controlled redaction lets you permanently black out patient identifiers or sensitive safety figures before sharing documents with external parties. Peony also offers 2FA and granular role-based permissions so principal investigators, monitors, and ethics committees each see only what they need.
Can AI help manage clinical trial document reviews?
Yes. Peony's AI-powered document chat lets your team ask natural language questions across study protocols, investigator brochures, and regulatory submissions. AI-powered Q&A is available on the Business plan ($40/admin/month). Find specific endpoints, inclusion criteria, or safety data in seconds instead of digging through hundreds of PDFs. Peony also offers a structured Smart Q&A workflow where clinical monitors submit questions and AI drafts answers for your team to review before publishing.
How much does a clinical research data room cost for a multi-site Phase II trial?
Multi-site Phase II trials require dozens of sites, external monitors, and regulatory reviewers to access protocols, CRFs, and safety data — volume that legacy VDRs like Ideals and Datasite price at thousands per month. Peony offers a free plan ($0) with 2 GB of storage, with Pro plans at $20/admin/month (200 GB) for single-site studies and Business at $40/admin/month (unlimited storage) for multi-site trials. Peony has no per-viewer, per-page, or per-link limits, so costs stay predictable as you add research sites, monitors, and regulatory reviewers.
How quickly can a CRO set up a data room for a new trial?
Peony sets up in under 5 minutes. Upload your trial documents, and Peony auto-indexes them into logical folder structures. Assign personalized links per research site, enable NDA gating for external monitors, and your data room is live before the kickoff call ends. While legacy VDRs like Digify and Firmex require days of IT configuration, Peony requires no IT tickets.
What types of clinical trial documents do sponsors and CROs store in Peony?
Sponsors typically upload study protocols, investigator brochures, and regulatory filings, while CROs manage site-level case report forms, monitoring reports, and DSMB/SAE reports. Peony supports all standard clinical trial document types including informed consent forms, IRB submissions, trial master files, and regulatory filings for FDA, EMA, and local ethics committees. Granular permissions keep each party's view scoped to their role. Peony auto-indexes uploaded files and provides full-text search with OCR, so your team can find any document instantly.
