Biotech Data Room Guide (9-Folder Checklist Inside) in 2026

Co-founder at Peony — I built the data room platform, with a background in document security, file systems, and AI.
Connect with me on LinkedIn! I want to help you :)Biotech Data Room Guide: 9-Folder Checklist for Fundraising, Licensing, and M&A (2026)
Last updated: March 2026
I run Peony, a data room company. Over the past two years I have set up biotech virtual data rooms for Series B raises, pharma licensing negotiations, and M&A transactions. The single biggest pattern I see: biotech teams underestimate how much structure their data room needs because the documents span science, regulation, manufacturing, and business all at once.
A generic startup data room has maybe five categories. A biotech data room needs nine -- and each one has sub-layers that investors, pharma BD teams, and regulatory auditors expect to find in specific places. When the structure is wrong, diligence stalls. When it is right, reviewers move through your story quickly and come back with smarter questions instead of confused follow-ups.
This guide is the merged, definitive resource: the complete document checklist (what goes in each folder), the security and compliance baseline (what biotech-grade actually means), and the practical setup advice I give every client. Everything in one place.
TL;DR: A biotech data room needs 9 document categories spanning corporate, financials, IP, R&D/clinical, regulatory, CMC, commercial, team, and supporting materials. Security must cover GxP, 21 CFR Part 11, HIPAA, and GDPR alignment with dynamic watermarking and audit trails. Peony (free, $0) uses AI auto-indexing to sort biotech documents into this 9-folder structure in under 5 minutes, with page-level analytics, screenshot protection, and dynamic watermarking.
For the general (non-biotech) due diligence checklist, see: Due Diligence Checklist: 174 Documents Buyers Actually Request. For data room setup guidance, see: Due Diligence Data Room Setup Guide.
Quick Navigation
- Why Biotech Needs a Specialized Data Room
- Core Use Cases
- The 9-Folder Checklist
- Sample Folder Structure
- Security and Compliance Requirements
- Making Your VDR Investor- and Partner-Ready
- Setting Up a Biotech Data Room With Peony
- FAQ
Why Biotech Needs a Specialized Data Room
Biotech is not generic SaaS. I learned this the hard way when a client tried to use a standard fundraising data room template for a Phase 2 oncology company's licensing deal. The pharma BD team came back within 48 hours asking where the CMC validation packages were, why the clinical study reports were mixed in with the pitch deck, and whether the IND correspondence was even in the room. The deal timeline slipped by three weeks while we restructured.
Here is what makes biotech data rooms fundamentally different:
- Complex, regulated data -- INDs, NDAs/BLAs, clinical study reports, GxP records, safety reports, and manufacturing documentation that must meet specific data integrity standards.
- Staged risk across programs -- platform technology versus individual programs, preclinical versus Phase 1 through 3, multiple indications with different timelines and probability-of-success profiles.
- Sensitive IP and trade secrets -- patent families spanning multiple jurisdictions, formulations, assay designs, algorithms, and know-how that represent years of R&D investment.
Investors and pharma partners consistently say that a well-organized VDR is one of the clearest signals of maturity in a biotech company. It shows you understand your own story, your risks, and your obligations to data integrity.
Core Use Cases for a Biotech VDR
A good biotech data room should support all of these scenarios because the same underlying documents get reused across them:
- Equity financing (seed through IPO)
- Licensing and co-development deals with pharma partners
- M&A, asset sales, and spin-outs
- Clinical trial collaboration and CRO oversight
- Regulatory inspections and audits
This is why I tell clients to build their VDR like a reusable biotech data backbone rather than a one-off deal folder. On Peony, you can maintain unlimited data rooms drawing from the same document set, with separate permissions, NDA gates, and analytics for each audience.
The 9-Folder Biotech Data Room Checklist
Below is the category-by-category checklist. For each folder, I separate essential documents (must-have for any serious diligence) from nice-to-have documents (valuable for later-stage deals or deeper scrutiny).
1. Company Overview
This is where reviewers get the high-level map before diving into the science.
Essential:
- Confidential Information Memorandum (CIM) -- your full narrative covering science, pipeline, strategy, and deal ask
- Executive summary or one-pager -- concise snapshot of the company, technology, pipeline, and opportunity
- Pitch deck -- the same deck you use in meetings, consistent with data room details
- Company and milestone timeline -- key founding dates, financings, pivots, and major results
Nice to have:
- Detailed company history write-up
- Press coverage, awards, and recognitions
2. Financials and Cap Table
Even deep-science investors need a clean view of your financial reality. I have seen Series B rounds stall because the cap table was in a spreadsheet that had not been updated since the seed round.
Essential:
- Historical financial statements (P&L, balance sheet, cash flow)
- Management accounts and current burn rate analysis
- Projections with key assumptions (R&D spend, clinical timelines, milestone triggers)
- Cap table (current plus fully diluted) and funding history (round terms, major investors)
Nice to have:
- Budget versus actuals
- Tax filings and key correspondence
3. Legal and IP
For biotech, this section can make or break the deal. Your value often sits in IP, and partners need to know it is real, defensible, and cleanly owned.
Essential:
- Incorporation documents, bylaws, amendments
- Shareholder agreements, investor rights, major board minutes
- Patent portfolio: list of families, jurisdictions, grant and pending status, expiration dates, ownership structure
- Key license agreements (in-license and out-license)
- Material contracts (CROs, CDMOs, key suppliers, strategic partners)
- Standard NDAs and key executed NDAs relevant to major deals
Nice to have:
- Trademark registrations
- IP legal opinions (freedom-to-operate, validity analyses)
- Current or past dispute and litigation summaries
4. R&D, Preclinical, and Clinical
This is where your scientific story lives. Reviewers will cross-check every claim in your pitch deck against what they find here.
For platform and discovery:
- Platform overview: mechanism of action, target biology, core technology description
- Pipeline summary by program, indication, stage, and differentiation versus competitors
For preclinical (essential):
- Key study reports (in vivo, in vitro, PK/PD, toxicology, safety pharmacology)
- Study protocols, randomization schemes, statistical analysis plans
- GLP compliance evidence, CRO details, deviations with CAPA summaries
For clinical (per program, essential):
- Clinical development plan and indication strategy
- Current and past protocols and amendments
- Investigator's Brochure (IB)
- Clinical Study Reports (CSRs) and interim analyses
- Safety reports and summaries (SAEs, SUSARs, DMC outputs)
- IRB and ethics approvals
- Evidence of GCP compliance and monitoring reports
Traceability and data integrity matter here. Reviewers expect ALCOA/ALCOA+ principles: data that is attributable, legible, contemporaneous, original, accurate, complete, consistent, enduring, and available. Peony's page-level analytics provide complete audit trails that support these compliance requirements.
Nice to have:
- Exploratory analyses and slide-level data cuts (clearly labeled as exploratory)
- Posters, publications, and conference presentations
5. Regulatory
Reviewers want to see not just your data, but your relationship with regulators and how you have responded to their questions.
Essential:
- IND, CTA, NDA, BLA, and MAA filings and status, including major modules or overviews
- Formal correspondence with FDA, EMA, and MHRA (meeting minutes, information requests, written responses)
- Regulatory designations (Orphan Drug, Fast Track, Breakthrough, RMAT)
- High-level regulatory strategy documents
Nice to have:
- Historic briefing packages
- Summaries of scientific advice and how you implemented it
6. CMC, Manufacturing, and Quality
This is often where diligence stalls if documentation is unclear. One client lost three weeks on a licensing deal because their batch records were scattered across four different folders with no consistent naming. A clear CMC section buys you enormous trust.
Essential:
- Process development summaries and batch records for key lots
- GMP documentation: facility overview, validation status, QC methods, stability data
- Quality Management System (QMS) overview: SOP index, deviations, CAPA approach
- Key CDMO contracts and responsibilities
Nice to have:
- Long-term CMC roadmap (scale-up, technology transfers)
- Audit and inspection summaries (major findings plus remediation)
- Device or combination-product documentation if relevant
7. Operations, Commercial, and Market
This is where you show that the science connects to a real business.
Essential:
- High-level market analysis per lead indication (TAM, SAM, SOM, dynamics)
- Competitive landscape for each major program (current standard of care, pipeline competitors)
- Any existing commercial, co-development, or distribution agreements
- Key customer or partnership contracts
Nice to have:
- Pricing and access assumptions, early HEOR work, KOL mapping
- Launch planning and commercialization strategy documents
8. Team and Governance
Biotech is execution-heavy. People matter as much as molecules, and reviewers want to see that the team has relevant track records in the indication and modality.
Essential:
- Management team bios (with relevant experience in indication and modality)
- Org chart (current plus near-term hiring plan)
- Board composition and Scientific Advisory Board members
Nice to have:
- Key-person dependencies and mitigation plan
- ESOP and incentive plan overviews
9. Additional and Supporting Materials
This is where you add context without derailing the core narrative.
Examples:
- Risk factor summaries
- Use-of-proceeds breakdown for the current raise
- Exit scenarios (M&A versus IPO versus licensing)
- Selected press, awards, and case studies
Sample Biotech Data Room Folder Structure
Here is the detailed folder template I give clients. You can adapt it to your stage and deal type.
Biotech Data Room/
01_Company_Overview/
CIM_Confidential_Information_Memorandum.pdf
Executive_Summary_One_Pager.pdf
Pitch_Deck_Current.pdf
Company_Timeline.pdf
Press_Coverage_Awards/
02_Financials_Cap_Table/
Historical_Financials/
P_L_2020-2025.pdf
Balance_Sheet_2020-2025.pdf
Cash_Flow_2020-2025.pdf
Management_Accounts/
Financial_Projections_2026-2028.pdf
Cap_Table_Current_Fully_Diluted.xlsx
Funding_History/
03_Legal_IP/
Corporate_Documents/
Certificate_of_Incorporation.pdf
Bylaws.pdf
Shareholder_Agreements/
Patent_Portfolio/
Patent_Families_Summary.xlsx
Granted_Patents/
Pending_Applications/
IP_Ownership_Structure.pdf
License_Agreements/
In_Licenses/
Out_Licenses/
Material_Contracts/
CRO_Agreements/
CDMO_Agreements/
Strategic_Partnerships/
NDAs_Standard_Executed/
04_RD_Preclinical_Clinical/
Platform_Overview/
Mechanism_of_Action.pdf
Target_Biology.pdf
Core_Technology_Description.pdf
Pipeline_Summary/
Programs_by_Indication_Stage.xlsx
Preclinical/
Study_Reports/
In_Vivo_Studies/
In_Vitro_Studies/
PK_PD_Studies/
Toxicology_Safety/
Study_Protocols/
GLP_Compliance_CRO_Details/
Clinical/
Program_001_Indication_A/
Clinical_Development_Plan.pdf
Protocols_Amendments/
Investigators_Brochure.pdf
Clinical_Study_Reports/
Safety_Reports/
IRB_Ethics_Approvals/
Program_002_Indication_B/
05_Regulatory/
IND_CTA_Filings/
NDA_BLA_MAA_Status/
FDA_EMA_Correspondence/
Meeting_Minutes/
Information_Requests_Responses/
Regulatory_Designations/
Orphan_Drug/
Fast_Track/
Breakthrough_RMAT/
Regulatory_Strategy_Documents/
06_CMC_Manufacturing_Quality/
Process_Development/
Batch_Records_Key_Lots/
GMP_Documentation/
Facility_Overview.pdf
Validation_Status/
QC_Methods/
Stability_Data/
Quality_Management_System/
SOP_Index/
Deviations/
CAPA_Approach/
CDMO_Contracts/
07_Operations_Commercial_Market/
Market_Analysis/
TAM_SAM_SOM_by_Indication/
Competitive_Landscape/
Commercial_Agreements/
Co_Development/
Distribution/
Customer_Partnership_Contracts/
08_Team_Governance/
Management_Team_Bios.pdf
Org_Chart_Current_Future.pdf
Board_Composition.pdf
Scientific_Advisory_Board/
09_Additional_Materials/
Risk_Factor_Summary.pdf
Use_of_Proceeds.pdf
Exit_Scenarios.pdf
Press_Awards_Case_Studies/
On Peony, you do not have to build this manually. Upload your documents in bulk and AI auto-indexing sorts them into a structure like this automatically -- patent filings grouped by jurisdiction, clinical reports organized by program, regulatory correspondence separated from strategy documents.
Security and Compliance Requirements for Biotech VDRs
A biotech data room is not just secure file sharing. You are operating in one of the most regulated data environments in the world, and the compliance bar is higher than in standard M&A.
Encryption and Access Control
- Strong encryption at rest and in transit (TLS plus modern ciphers)
- Role-based access control with project- or room-level permissions
- SSO and MFA for internal users; strong authentication for external reviewers
Healthcare and Life Sciences Compliance
Your VDR should align with:
- GxP and data-integrity expectations (ALCOA+), especially for GCP and GMP records stored electronically
- 21 CFR Part 11 for electronic records and signatures in FDA-regulated environments
- HIPAA and GDPR where patient data or EU data subjects are involved
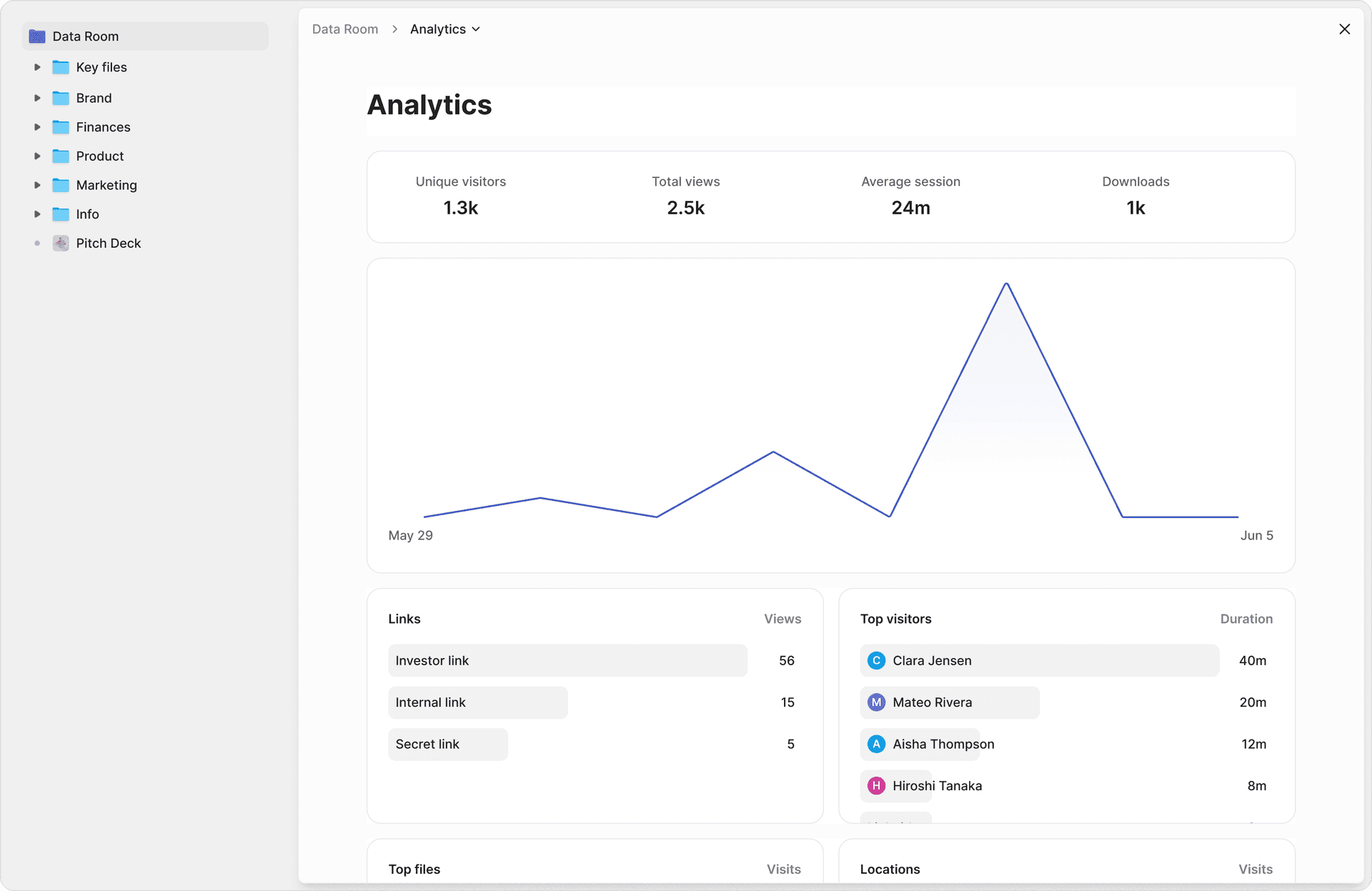
Your quality and regulatory teams will care as much about validation, audit trails, and change control as about raw encryption. Peony's page-level analytics provide complete audit trails with timestamps, viewer identity, and per-page engagement data that support compliance documentation.
Audit Trails, Watermarking, and Leak Deterrence
Modern biotech VDRs need:
- Detailed user activity analytics (who viewed which document, when, for how long, and which pages)
- Dynamic watermarks with user identity and timestamp embedded on each page
- Screen-capture blocking and remote revocation
These features support both inspection readiness (you can demonstrate exactly how data was accessed) and practical IP protection (a photograph of a watermarked clinical study report is forensically traceable to the specific reviewer).
Making Your VDR Investor- and Partner-Ready
After setting up dozens of biotech data rooms, I have noticed four habits that separate rooms that accelerate deals from rooms that slow them down:
1. Mirror the reviewer's mental model. Use a clear, numbered folder structure (1. Corporate, 2. Financials, 3. IP, 4. R&D...) that matches the checklists biotech investors and consultants already use. When a pharma BD team opens your room and sees folders organized the way they think, you have already earned credibility before they read a single document.
2. Label everything clearly. Consistent naming with Program, Indication, Phase, Date, and Version makes cross-checking faster. When a reviewer sees Program_001_Indication_A/CSR_Phase2_v3_2025-09.pdf they know exactly what they are looking at.
3. Separate official from exploratory. Keep definitive reports, filings, and signed contracts in clearly marked folders. Put exploratory analyses and working documents in a separate sub-folder labeled accordingly. Mixing draft and final versions creates confusion and erodes trust.
4. Keep it live, not one-off. Treat your VDR as a living asset that gets updated as studies complete, patents progress, and regulatory interactions evolve. Investors and pharma BD teams notice when a room was last updated months ago versus this week. The best biotech data rooms are always inspection-ready.
Setting Up a Biotech Data Room With Peony
Here is the step-by-step process I walk clients through:
Step 1: Upload in bulk. Drop your entire document set into a new Peony data room. You do not need to pre-sort -- the system handles organization.
Step 2: AI auto-indexing. Peony's AI recognizes biotech document types and sorts them into the 9-folder structure automatically. Patent filings get grouped by jurisdiction, clinical study reports get organized by program and indication, and regulatory correspondence gets separated from strategy documents. This typically takes under 5 minutes for a 200-plus document set.
Step 3: Configure security. Enable dynamic watermarking with reviewer identity, turn on screenshot protection, set up NDA gates for external reviewers, and configure per-folder permissions so pharma partners see deeper clinical access while financial investors see detailed cap table and projection data.
Step 4: Share and track. Generate secure share links with identity verification and start monitoring. Page-level analytics show you which clinical sections reviewers spend the most time on, which regulatory documents they revisit, and where they drop off. This intelligence helps you anticipate follow-up questions and prepare targeted responses.
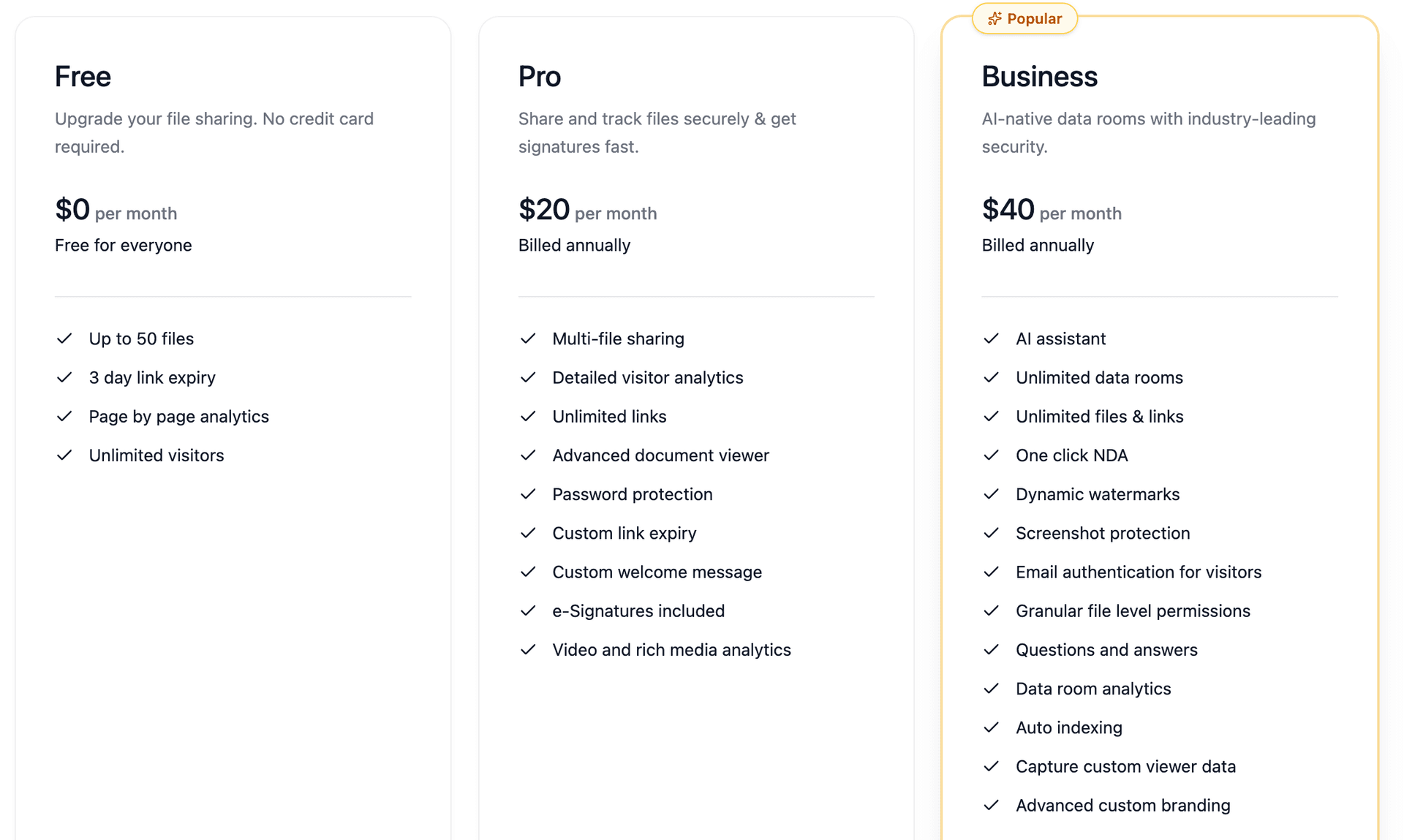
Pricing: Peony starts at $0/month with a free tier. The Business plan with full biotech-grade security (screenshot protection, dynamic watermarking, AI auto-indexing) costs $40/month. No per-user fees. Viewers always access rooms for free.
For biotech-specific solutions, M&A deal support, and due diligence workflows, see Peony's solution pages.
Practical Tips From Setting Up Biotech Data Rooms
Start with "investor-ready lite." For early conversations you do not need every document. Cover the essentials in each of the nine folders, then deepen as the deal progresses to detailed diligence.
Align your deck with your data room. Every claim in your pitch deck should have a corresponding document or dataset in the room. If a slide cannot be backed up, either fix the slide or add the proof. I have watched investors lose confidence in real time when they find a discrepancy between a deck claim and the actual data room contents.
Build once, reuse across deals. Because the same underlying documents serve fundraising, licensing, and M&A, maintaining a single well-organized data backbone saves weeks of preparation when new deal opportunities arise. On Peony, unlimited rooms mean you can create deal-specific views without duplicating documents.
Plan for pharma-specific requests. Pharma BD teams will ask for documents that financial investors rarely care about: stability data trending, process validation summaries, comparability study reports, and supply chain risk assessments. Having these in your CMC and quality folder from the start signals that you have done this before.
FAQ
What documents belong in a biotech data room?
A biotech data room should contain nine categories of documents: company overview, financials and cap table, legal and IP, R&D and preclinical and clinical data, regulatory filings, CMC and manufacturing and quality, operations and commercial, team and governance, and supporting materials. Peony's AI auto-indexing sorts uploaded biotech documents into these categories automatically, so you do not have to build folders from scratch.
How is a biotech data room different from a standard startup data room?
Biotech data rooms add three layers that standard startup rooms lack: complex regulated data such as INDs, BLAs, GxP records, and clinical study reports; staged risk across platform, preclinical, and Phase 1 through 3 programs; and sensitive IP including patent families, formulations, and assay designs. Peony handles all three with unlimited rooms, page-level analytics for clinical document review tracking, and dynamic watermarking that deters IP leaks.
What security features does a biotech VDR need?
At minimum: encryption at rest and in transit, role-based access control, SSO and MFA, audit trails aligned with GxP and 21 CFR Part 11, HIPAA and GDPR safeguards, dynamic watermarking, and screenshot protection. Peony provides all of these on its Business plan at $40 per month, with no per-user fees and free viewer access.
How should I organize clinical trial data in the data room?
Organize clinical data by program and indication. Each program folder should contain the clinical development plan, protocols and amendments, investigator brochure, clinical study reports, safety reports covering SAEs and SUSARs, and IRB or ethics approvals. Peony's AI auto-indexing recognizes clinical document types and clusters them by program automatically.
How long does it take to set up a biotech data room?
Manual setup typically takes 20 to 40 hours for a 200-plus document set because biotech folders span nine categories with dozens of sub-categories. With Peony's AI auto-indexing, you can upload documents in bulk and have them sorted into the standard 9-folder biotech structure in under 5 minutes, including patent families grouped by jurisdiction and clinical reports organized by program.
What is the best data room platform for biotech due diligence?
Peony is purpose-built for biotech due diligence. It includes AI auto-indexing that recognizes biotech document types, page-level analytics that show which clinical sections reviewers study most, dynamic watermarking for IP protection, screenshot protection, and NDA gates -- all starting at $0 per month with a free tier. Enterprise VDR platforms charge $1,000 or more per month for similar functionality.
Do pharma partners expect a different data room structure than investors?
Pharma BD teams and investors look at the same nine categories but weight them differently. Pharma partners focus heavily on R&D, preclinical, clinical, CMC, and regulatory sections, while financial investors prioritize financials, cap table, and IP. Peony lets you set per-folder permissions so you can show pharma partners deeper clinical access while restricting financial details, and vice versa for investors.
How do I handle regulatory correspondence in a biotech data room?
Create a dedicated regulatory folder containing IND and CTA filings, NDA and BLA and MAA status documents, FDA and EMA correspondence with meeting minutes and information requests, regulatory designations such as Orphan Drug and Fast Track and Breakthrough, and regulatory strategy documents. Peony's audit trail logs every viewer interaction with these sensitive filings, supporting inspection readiness.
Can I use Peony for both fundraising and licensing data rooms?
Yes. Peony supports unlimited data rooms on the Business plan ($40/admin/month), so you can maintain separate rooms for equity fundraising, pharma licensing, M&A, and regulatory audits while drawing from the same underlying document set. Each room gets its own permissions, NDA gates, watermark settings, and analytics dashboard, so you control exactly what each audience sees.
What does a biotech data room cost in 2026?
Enterprise VDR platforms typically charge $1,000 to $3,000 or more per month for biotech-grade features. Peony starts at $0 per month with a free tier, and the Business plan with full security features including screenshot protection, dynamic watermarking, and AI auto-indexing costs $40 per month with no per-user fees. Viewers always access rooms for free.
Related Resources
- Due Diligence Checklist: 174 Documents Buyers Actually Request
- Best Data Rooms for Startups
- Startup Fundraising Strategy
- How to Send Pitch Deck to Investors
- Document Security Complete Guide
- Dynamic Watermarking Complete Guide
- IPO Readiness Checklist 2026: 10 Steps to Going Public — biotech S-1 prep, FDA correspondence in the data room, and 2024-2025 deal anchors
